 Elements can be classified as metals, metalloids, and nonmetals, or as a main-group elements, transition metals, and inner transition metals. Elements in the same group of the periodic table have similar chemical properties. 2.3: The Periodic Table The discovery of the periodic recurrence of similar properties among the elements led to the formulation of the periodic table, in which the elements are arranged in order of increasing atomic number in rows known as periods and columns known as groups.An amu is defined as exactly \(1/12\) of the mass of a carbon-12 atom and is equal to 1.6605 \(\times\) 10−24 g. The mass of one atom is usually expressed in atomic mass units (amu), which is referred to as the atomic mass. The nucleus contains protons and neutrons its diameter is about 100,000 times smaller than that of the atom. 2.2: Atomic Structure and Symbolism An atom consists of a small, positively charged nucleus surrounded by electrons.Rutherford’s gold foil experiment showed that atoms have a small, dense, positively charged nucleus the positively charged particles within the nucleus are called protons. Millikan discovered that there is a fundamental electric charge-the charge of an electron.  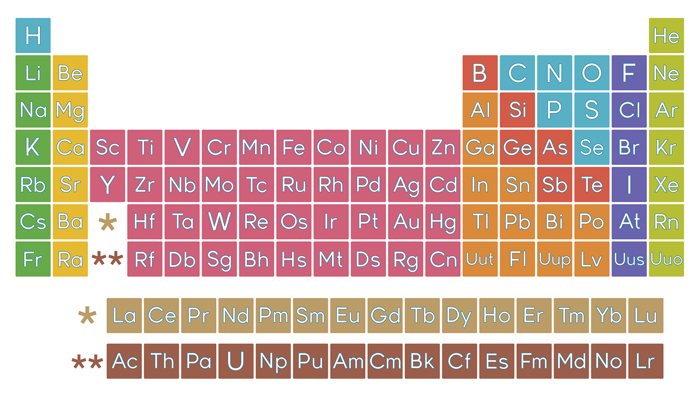 
Thomson’s cathode ray tube showed that atoms contain small, negatively charged particles called electrons. 2.1: Evolution of Atomic Theory Although no one has actually seen the inside of an atom, experiments have demonstrated much about atomic structure. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
